祝贺李守湖博士的文章在ACS Applied Materials & Interfaces上发表
更新时间:2021-04-24
文章题目:Iodine-Substituted Lithium/Sodium closo-Decaborates: Syntheses, Characterization, and Solid-State Ionic Conductivity
作者:Shouhu Li, Pengtao Qiu*, Jiaxin Kang, Yiming Ma, Yichun Zhang, Yigang Yan, Torben R. Jensen, Yanhui Guo, Jie Zhang*, and Xuenian Chen*
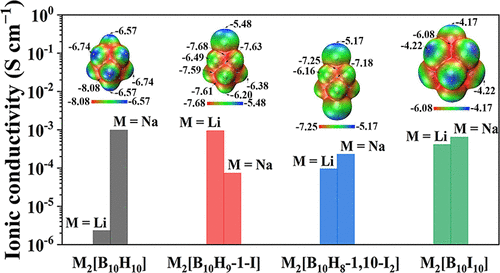
Solid-state electrolytes based on closo-decaborates have caught increasing interests owing to the impressive room-temperature ionic conductivity, remarkable thermal/chemical stability, and excellent deformability. In order to develop new solid-state ion conductors, we investigated the influence of iodine substitution on the thermal, structural, and ionic-conduction properties of closo-decaborates. A series of iodinated-closo-decaborates, M2[B10H10−nIn] (M = Li, Na; n = 1, 2, 10), were synthesized and characterized by thermal analysis, powder X-ray diffraction, and electrochemical impedance spectroscopy; the stability and ionic conductivity of these compounds were studied. It was found that with the increase of iodine substitution on the closo-decaborate anion cage, the thermal decomposition temperature increases. All M2[B10H10−nIn] exhibit amorphous structure. The ionic conductivity of Li2[B10H10−nIn] is higher than that of the Li2[B10H10] parent compound. An ionic conductivity of 2.96 × 10−2 S cm−1 with an activation energy of 0.23 eV was observed for Li2[B10I10] at 300 ℃, implying that iodine substitution can improve the ionic conductivity. However, the ionic conductivity of Na2[B10H10−nIn] is lower than that of Na2[B10H10], and increases with the increase of iodine substitution, which could be associated with the increase of electrostatic potential, mass, and volume of the iodinated anions. Moreover, Li2[B10I10] offers a Li-ion transference number of 0.999 and an electrochemical stability window of 3.3 V including good compatibility with Li anode, demonstrating the potential application in high-temperature batteries.
期刊年卷期页码:ACS Appl. Mater. Interfaces 2021, 13, 17554-17564
链接地址:https://pubs.acs.org/doi/10.1021/acsami.1c01659